Special Report: Complementary Alternative and Integrative Medicine Can Help Meet Challenges of COVID-19


The COVID-19 pandemic has unmasked domestic and global deficits and systemic inequities in mental health care. The additive burden of losses and stresses has caused rates of anxiety, depression, and posttraumatic stress to rise among the general public and the health care workforce.
In this article, we review three evidence-based complementary alternative and integrative medicine (CAIM) options that have been the most useful for improving emotional resilience during the COVID-19 crisis. These are Voluntarily Regulated Breathing Practices, Rhodiola rosea, and S-adenosylmethionine.
These three CAIM treatments alone or in combination with each other and/or psychotropic medications are potentially beneficial for a wide range of disorders. Moreover, physicians and other health care professionals who use these modalities themselves find that they have more energy, better mood, a sharper mind, less burnout, and better physical health.

Table 1
More physicians are taking the time to study CAIM; however, the number who are qualified and available for expert consultations is quite limited. Consequently, psychiatrists cannot rely on referring patients to CAIM specialists, but instead they need to educate themselves regarding how to use these treatments safely and effectively (see Table 1). Those who do will be rewarded with a new set of skills for improving patient outcomes; reducing the burden of medication side effects; and maintaining their own capacities, including their will to serve patients during these challenging times.
Breath in the Time of COVID-19
The COVID-19 epidemic has spawned secondary epidemics of stress, anxiety, depression, and posttraumatic stress disorder (PTSD). Voluntarily Regulated Breathing Practices (VRBPs), particularly when combined with gentle movement, have been shown to improve anxiety, mood, trauma, attention, cognitive function, energy, and social engagement. While intense breath practices tend to increase energy, slow breath practices, such as Coherent Breathing (also called Resonant Breathing) at 4.5 to 6.0 breaths a minute, rapidly reduce anxiety, hyperarousal, and defensiveness. Coherent Breathing promotes autonomic balance by downregulating overactivity of the sympathetic nervous system and upregulating the parasympathetic system, which is underactive in anxiety disorders, depression, PTSD, and chronic fatigue.
In addition to balancing the activity of the autonomic system, preliminary evidence suggests that Coherent Breathing may increase levels of the inhibitory neurotransmitter gamma-aminobutyric acid (GABA) in circuits involved in emotion regulation. For example, in a trial of patients with major depressive disorder who practiced yoga and Coherent Breathing for 12 weeks, Chris Streeter, M.D., and colleagues reported significant improvement in depression and suicidal ideation, as well as a significant increase in thalamic GABA levels on mass resonance spectroscopy. The study appeared in the March 2020 Journal of Alternative and Complementary Medicine.
Interoception is the perception of feelings from inside the body. With each breath, millions of sensors throughout the respiratory system send afferent messages to central regulatory structures involved in emotion regulation, stress response, cognitive function, and behavior. These include the amygdala, hypothalamus, thalamus, insular cortex, prefrontal cortex, and anterior cingulate cortex. In contrast to automatic breathing, intracranial iEEG recordings of VRBPs showed increased “breath coherence,” entrainment of electrical activity with the breath pattern in critical brain areas: insula; amygdala; and premotor, olfactory, and caudal-medial frontal (executive function) and temporal cortices. Furthermore, when subjects focused attention on breathing, iEEG-breath coherence increased in anterior cingulate premotor, insular, and hippocampal cortices. In a magnetoencephalography study in humans, mental regulation of slow VRBPs modulated cortical alpha activity in a well-structured patten of alpha waves over wide areas of the brain.
In accord with the Polyvagal Theory, slow VRBPs can activate afferent myelinated vagal parasympathetic pathways, which may reduce defensive reactions; facilitate a psychophysiological state of feeling safe; activate the social engagement network; and increase feelings of love, connectedness, empathy, and cooperation.
Breath practices can be taught efficiently to individuals, families, or groups in any clinical setting or remotely online. Learning how to teach the practices and how to observe the patient’s breath pattern is important to assure the optimal performance of breath techniques. The usual “dose” for coherent breathing is 20 minutes a day and as needed for anxiety, sleep, or pain. For severe anxiety or PTSD, 20 minutes twice daily and as needed for stress/anxiety are advised.
Breath-Based Program for Pandemic-Related Stress in Staff Working With Special Needs Children
Agencies responsible for children, especially those with special needs, have been under severe, prolonged COVID-19-related stress in the United States and other countries. Contributing factors include disrupted treatment, staff shortages, family dysfunction, and emotional/behavioral deterioration among the children. Worsening of the child’s symptoms and family strain create increased demand for services.
In a December 2020 pilot study conducted by two of the authors (Dr. Gerbarg and Dr. Brown), 24 members of the staff of the Regional Integrative Support for Northern Ireland (RISE NI) completed a live 12-hour online program (4 hours a day for three consecutive days) of breathing, movement, and awareness practices called Breath-Body-Mind (BBM). The researchers were encouraged to do the practices at home 20 minutes a day and attend a weekly 45-minute group practice. Standardized measures of anxiety and stress were obtained before the program, immediately after, and at week 6. The group mean score on the Stress Overload Scale-Short was well above the normative level at baseline, declined significantly immediately after BBM, and fell significantly below the norm after six weeks of practice. A large proportion of the participants reported moderate to very strong improvements, including less worry (70%), clearer mind (79%), more energy (78%), and more empathy (62%).
Breath-Based Programs for Maternal, Fetal, and Newborn Stress
Maternal distress can adversely affect children’s development before as well as after birth. The rates of anxiety and depression during the COVID-19 epidemic among pregnant women are estimated to have doubled or tripled. Maternal depression and anxiety are associated with increased risk of preterm birth, lower birth weight, postpartum depression, and behavioral difficulties in children.
Slow, gentle breath practices, such as Coherent Breathing at 5 to 6 breaths a minute with equal inhalation and exhalation, are a safe way to reduce anxiety and increase maternal heart rate variability (HRV), which is associated with increased fetal HRV. Coherent Breathing with visualization has been used to calm anxious high-risk mothers and agitated premature infants in a neonatal care setting, facilitating positive coregulation and mother-infant bonding. This can also reduce regurgitation during feedings, which is essential for weight gain and eventual discharge.
Herbal Adaptogens: Best in Show
Among the adaptogenic herbs, Rhodiola rosea (R. rosea) has the greatest potential for use in psychiatric practice. A substantial evidence base of safety and efficacy supports the use of this phytomedicinal to enhance mood, energy, and cognitive function. Adaptogens contain numerous bioactive substances that protect organisms from damage by infections, chemical toxins, free radicals, radiation, cancer, stress, extreme heat and cold, low oxygen conditions, and other hazards. As a metabolic regulator, R. rosea increases mitochondrial production of energy transport adenosine triphosphate (ATP) and supports repair of cellular components. These fundamental cellular effects contribute to numerous benefits in chronic fatigue, depression, perimenopausal depression, attention-deficit/hyperactivity disorder, PTSD, and cognitive dysfunction.
A review of 11 randomized, controlled trials (RCTs) concluded that R. rosea may have beneficial effects on physical performance, mental performance, and mental illness. A pharmacological review noted that R. rosea extracts and one of the constituents, salidroside, demonstrated anti-aging, anti-inflammatory, immunostimulant, DNA reparative, and anti-cancer effects.
The R. rosea root contains compounds with multiple mechanisms of action throughout the body. The methods of cultivation and extraction of these compounds are critical to the quality of products. Clinicians can identify high-quality products by using those proven to be effective in published studies and those recommended by experienced CAIM practitioners. (See the book How to Use Herbs, Nutrients, and Yoga in Mental Health Care by the authors of this article.)
R. rosea for Chronic Fatigue
R. rosea can improve mental and physical energy in patients with chronic fatigue syndrome following infections (for example, Lyme disease and “long” COVID-19); medication-induced fatigue; and fatigue associated with cancer, medical illnesses, or depression. For example, in an open-label multicenter clinical trial, 100 subjects with prolonged or chronic fatigue syndrome symptoms were administered an ethanol extract of R. rosea. The greatest change was observed after one week of treatment. Fatigue continued to decline with statistically significant improvement at week 8. The safety assessment was favorable, most adverse events being mild and unrelated to the extract.
R. rosea for Mental Fatigue
Mental fatigue causes a decline of mental functioning, often with an increased error rate. This has been associated with reduced “top-down” resources and decreased attention. Studies have shown that R. rosea can reduce mental fatigue, reaction time, and errors on tasks. A 12-week study of 50 healthy adults given 200 mg twice daily of R. rosea demonstrated significant improvements on a battery of attentional tasks at weeks 6 and 12 in indicators of processing resources, decreased reaction time, and reduced rate of errors.
R. rosea for Depression
R. rosea can be used to treat patients with mild-to-moderate depression. In a placebo-controlled study of 128 mild to moderately depressed patients, 150 mg TID of Arctic root (another name for R. rosea) was associated with significantly improved depression in two-thirds of subjects compared with placebo. A double-blind, randomized, controlled study using Arctic root SHR-5 as a monotherapy for 89 adults with mild to moderate depression (baseline score on the Hamilton Depression Rating Scale [HAM-D] ranging from 12 to 31) found that those given Arctic root SHR-5 (340 mg/day or 680 mg/day) for six weeks showed significantly greater improvements in mean HAM-D scores compared with the placebo group.
R. rosea Augmentation of Antidepressants
The relative safety and efficacy of R. rosea capsules in combination with sertraline for mild to moderate major depressive disorder was confirmed in a 12-week double-blind, placebo-controlled trial published in 2020. One hundred patients were randomized to three groups, and all three showed significant improvements on measures of depression. Post-treatment declines in HAM-D, Beck Depression Inventory (BDI), and Clinical Global Improvement (CGI) scores were greater for group B (sertraline + Rhodiola at 600 mg/day) than for group A (sertraline + placebo) and group C (sertraline + Rhodiola 300 mg/day + placebo).
Two of the authors of this report (Drs. Gerbarg and Brown) find R. rosea particularly beneficial for depression in perimenopausal women with fatigue, “brain fog,” mild memory retrieval difficulties, and loss of libido. The observed improvements in energy, mood, mental sharpness, memory, and sexual desire may be related to activating effects and increased mitochondrial energy as well as its effects as a possible selective estrogen receptor modulator (SERM). SERMs are of interest because they may affect estrogen receptors differently, depending on the organ. The ideal SERM would activate estrogen receptors in the brain and bones but not in the breast, ovaries, or uterus. The net result would be preservation of brain function and bone structure without increased risk of cancer.
Attention-Deficit Disorder With or Without Hyperactivity
For patients with mild attention-deficit disorder with or without hyperactivity and those who cannot tolerate the side effects of stimulant medications at therapeutic doses, R. rosea is worth a trial. Prescription stimulants, whose pharmacological activity peaks and falls rapidly, can cause withdrawal symptoms (restlessness, jitteriness, anxiety, and insomnia) and may contribute to addiction. In patients whose stimulant medication wears off late in the day, a moderate midday dose of R. rosea can maintain attentional focus without disturbing sleep because the effect fades after 6 to 8 hours.
Stimulant abuse can lead to increased blood pressure and heart rate, blood vessel constriction, increased blood glucose, rapid or irregular heartbeat, delirium, panic, psychosis, paranoia, myocardial infarction, heart failure, or seizures. In contrast, the use of R. rosea causes no withdrawal symptoms or addiction, no loss of appetite, no cardiac disease, and no abnormal mental states. We find in clinical practice that R. rosea is generally safe in therapeutic doses and does not lead to abuse or withdrawal syndromes.
Effective Dosing of R. rosea
In clinical practice, R. rosea has been found to be an effective augmentation to antidepressant drugs, particularly for symptoms of fatigue, apathy, psychomotor retardation, anhedonia, memory impairment, and poor concentration. Best effects occur when taken on an empty stomach 30 minutes before breakfast and lunch, with total daily doses ranging from 300 mg/day to 900 mg/day. The use of best-quality R. rosea as monotherapy or augmentation for moderate to severe depression warrants further study.
Risks, Adverse Effects, and Potential Medication Interactions
In rat toxicity studies, the median lethal dose was calculated to be 28.6 ml/kg, approximately 3,360 mg/kg. Since the usual clinical doses are 200 mg/day to 900 mg/day, there is a considerable margin of safety. A randomized crossover study of 13 healthy volunteers found no effects on cytochrome P (CYP) enzymes except for modest inhibition of CYP2C9 (possibly significant for substrates with a narrow therapeutic index, such as phenytoin and warfarin).
As with prescription antidepressants, R. rosea may increase the risk of mania in bipolar patients. It can occasionally cause jitteriness, agitation, or insomnia in patients who are sensitive to stimulating agents and may have additive effects when combined with caffeine. Uncommon side effects include anxiety, headache, bruising (with doses of 900 mg or more a day), palpitations, increased blood pressure, and chest pain. Animal studies show no teratogenic effects.
Neutraceutical SAMe Acts as Natural Antidepressant
S-adenosylmethionine (SAMe), a central metabolite in animal cells, provides methyl groups and other moieties for production of neurotransmitters, lipid membranes, proteins, and glutathione. SAMe donates methyl groups (CH3) involved in activation and deactivation of histones that turn on and off the genes in DNA. It has been used to treat depression, osteoarthritis, liver toxicity, gallstones of pregnancy, Parkinson’s disease, and other medical conditions for over 30 years, mainly in Europe.
Extensive research and clinical experience confirm that SAMe has very few side effects and virtually no drug interactions. The adverse effects of prescription antidepressants, particularly weight gain and sexual dysfunction, contribute to patient discontinuation. Because SAMe does not cause sexual side effects, sedation, weight gain, or cognitive interference, it is more acceptable to patients.
An APA work group of the Council on Research reviewed 17 open-label trials; 19 double-blind, randomized, controlled trials; and 21 trials comparing SAMe with prescription antidepressants. The review, by Anup Sharma, M.D., Ph.D., et al., was published June 2017 in the Journal of Clinical Psychiatry. The researchers reported encouraging evidence of the efficacy and safety of SAMe as a monotherapy and as an augmentation for other antidepressants. Their review also identified “supportive early evidence for SAMe in certain neurocognitive, substance use, and psychotic disorders.” Additional clinical studies are needed to further delineate the role of SAMe in neuropsychiatric conditions.
Unsolved Mysteries of SAMe Research
SAMe is one of the most useful treatments available for children, and adults, including older people, pregnant women, and medically ill patients. We have used SAMe in our clinical practices for over 20 years to treat patients with depression, cognitive impairment, mental and physical fatigue, attention-deficit disorder, arthritis, and liver dysfunction. Nevertheless, several recent three-armed, well-designed, double-blind, randomized, controlled studies of patients with mild to moderate major depressive disorder found no significant differences in improvement among placebo, SAMe, and prescription antidepressants. Considering these reports, why do we continue to recommend SAMe for treatment of depression?
SAMe was approved for marketing as a nutraceutical for depression in the United States in 1999. Based on its review of SAMe studies, the U.S. Department of Health and Human Services Agency for Healthcare Research and Quality concluded that SAMe was equivalent to standard pharmacotherapy for depression and osteoarthritis with fewer side effects.
Prior to 2005, most studies used intramuscular SAMe or 800 mg of 1,4-butanedisulfonate SAMe twice daily by mouth in patients with moderate to severe depression. Eventually, a large pharmaceutical company bought the European company that manufactured high-quality butanedisulfonate SAMe and discontinued sales in the United States, but continued to sell it in other countries. Subsequently, American researchers studying SAMe for major depression used the tosylate or toluenate forms. Since then, several trials of SAMe for mild to moderate depression in this country have reported inconclusive results and large placebo-response rates, and some have used doses below the recommended dose of 1,600 mg/day.
Nonconclusive results from these three-armed, well-designed studies may be due to patient selection or heterogeneity (especially using patients with mild or nonbiological depression), the use of less potent forms of SAMe, or other unknown factors. Reanalysis of data from these studies has not yet identified the reasons for the high placebo-response rates and inconclusive data. The authors of one of these studies—David Mischoulon, M.D., Ph.D., et al.—deserve credit for writing that they continue to prescribe SAMe to their patients with good results, thereby acknowledging the discrepancy between the study data and their own clinical observations.
An independent manufacturer, ExtremeV, is now selling high-quality 1,4-butanedisulfonate SAMe in the United States under the brand name Azendus. In our clinical practices, we have found Azendus to be more potent and to require lower doses compared with other SAMe brands.
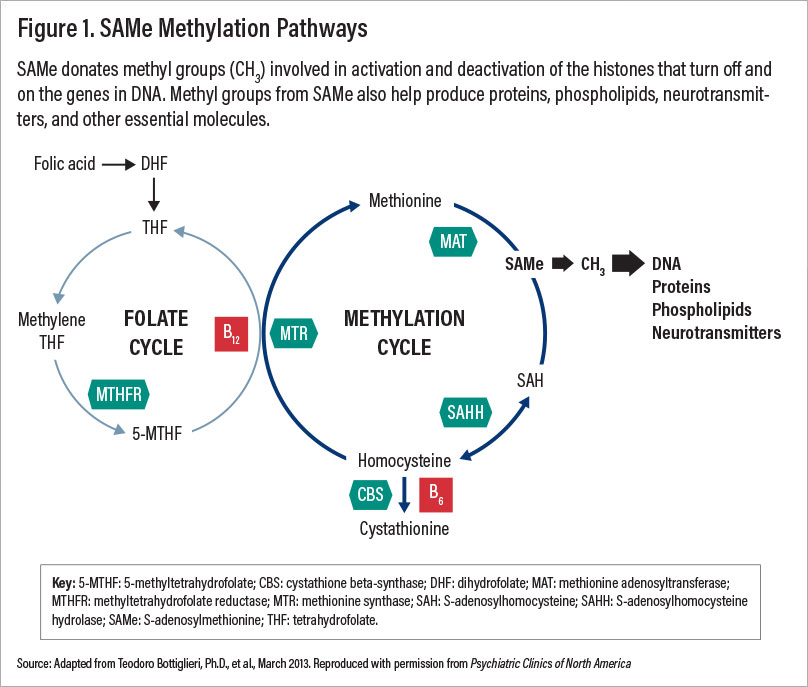
Figure 1
SAMe methylation pathways involved in production of neurotransmitters require the cofactors B12 (methylcobalamine) and folate (see Figure 1). Low levels of these cofactors are associated with suboptimal response to SAMe and other antidepressants. Adding 1,000 ug/day B12 and 800-1,000 ug folate can enhance the antidepressant effects of SAMe in patients with B12 or folate deficiencies.
SAMe for Treatment of Depression
For a review of SAMe research, see the article by the work group of APA’s Council on Research by Dr. Sharma. As an activating antidepressant, SAMe helps to energize depressed patients who have low energy, fatigue, low motivation, and hypersomnia. SAMe has demonstrated efficacy as a monotherapy for depression. Studies also support efficacy as an augmentation strategy with prescription antidepressants. Studies of SAMe augmentation have not shown adverse interactions with numerous antidepressants, including monoamine oxidase inhibitors.
Depression With Comorbid Disorders
Major depressive disorder is common in patients with rheumatoid arthritis, with a prevalence of 13% to 42%, at least double to four times the rate in the general population. A clinical review of SAMe identified 12 studies showing analgesic and anti-inflammatory effects in patients with osteoarthritis. In addition, six studies have shown that 800 mg to 1,200 mg of SAMe a day improved depression and pain in patients with fibromyalgia.
Depression in patients with hepatic disease may be primary, secondary to hepatic dysfunction, or due to medications. Alcohol abuse can deplete liver SAMe supplies. In patients with cirrhosis or hepatitis due to alcohol, drugs (including prescription drugs), toxins, infections, or gallstones (including during pregnancy), studies show that SAMe protects the liver, normalizes elevated liver function tests, increases survival, and delays the need for liver transplantation.
Carbidopa and levodopa (L-dopa) are commonly used to treat Parkinson’s disease. L-dopa depletes brain stores of SAMe. Depletion of SAMe probably contributes to depression in patients with Parkinson’s, which can be resistant to standard antidepressants. Two small studies of SAMe in Parkinson’s patients showed significant improvements in depression with doses ranging from 1,200 mg/day to 4,000 mg/day.
The rate of depression in people living with HIV is estimated to be 30% to 50%. SAMe deficiency occurs in HIV patients. In an open eight-week study of 20 patients with HIV and major depressive disorder, SAMe at 400 mg twice a day with folic acid (800 mcg/day) and B12 (1,000 mcg/day) was associated with significant improvement in depression by week 4.
SAMe During Pregnancy and Postpartum
As a natural metabolite, SAMe may be safer during pregnancy than synthetic antidepressants. For more than 20 years in Europe, SAMe has been used to treat women with gallstones during pregnancy. A meta-analysis of 10 RCTs by Fan Zhou and colleagues published in April 2014 using ursodeoxycholic acid and SAMe to treat intrahepatic cholestasis of pregnancy found a reduced rate of cesarean sections, preterm birth, and fetal asphyxia. Two follow-up studies of children of mothers who had taken SAMe for cholestasis during pregnancy found no evidence of abnormal physical or psychological development up to age 4.
In a double-blind, randomized, controlled trial, women with postpartum depressive symptoms who were given SAMe improved significantly more after 10 days of treatment than women who took placebo.
Measures of CSF SAMe in infants found concentrations that are three to seven times the CSF concentrations in adults. Consequently, it is unlikely that the transfer of SAMe through breast milk could cause harm. Studies of CAIM treatments for depression during pregnancy are warranted.
SAMe for Childhood Depression and Obsessive-Compulsive Disorder
Although there are no published controlled studies on SAMe for children, clinically it has been used successfully to treat children and adolescents for depression and obsessive-compulsive disorder (OCD). Three cases were reported of two sisters, aged 8 and 11, and one boy, aged 16. The parents did not want to expose their children to potential side effects of prescription medications. The girls had serious major depressive disorder, characterized by crying, withdrawal, sadness, and preoccupation with themes of death. The 11-year-old began 200 mg/day SAMe. At 600 mg/day, her depression remitted with no side effects. Her 8-year-old sister’s depression also resolved completely on 200 mg SAMe twice a day. In clinical practice, one of the authors of this article (Dr. Brown) treated four children and three adolescents for major depressive disorder with SAMe butanedisulfonate manufactured by Knoll Pharmaceutical Co. In all cases, the children could not tolerate any prescription antidepressants, and in all cases SAMe butanedisulfonate was well tolerated and effective. The youngest child responded well to SAMe 800 mg a day. One of the adolescents, who had both major depressive disorder and OCD, responded only to a high dose of SAMe—2,800 mg a day. Four more of this author’s (Dr. Brown’s) child patients with OCD responded very well to a 3- to 6-month course of SAMe. One of these, a 10-year-old boy with severe OCD, attained remission of symptoms after taking 2,000 mg oral SAMe butanedisulfonate (Azendus) every morning for 12 weeks. He had no side effects other than mild, tolerable gastrointestinal discomfort. Studies of SAMe butanedisulfonate for depression and OCD in children are much needed to support this low-risk treatment option.
SAMe Side Effects and Contraindications
SAMe should be taken on an empty stomach 30 to 60 minutes before breakfast and lunch to optimize absorption. Late afternoon or evening doses may interfere with sleep. For depressed patients with anxiety and agitation, it may be necessary to use a benzodiazepine or buspirone during the first few weeks to reduce the stimulating effects of SAMe. As the antidepressant action is established, anxiolytics can usually be tapered.
Mild nausea is the most common side effect of SAMe. This can usually be relieved by eating a light snack before taking SAMe, taking ginger capsules or tea if nausea occurs, or decreasing the dose of SAMe. Loose bowel movements, flatulence, or abdominal pain can also occur. Patients who are prone to nausea, gastric irritation, or irritable bowel symptoms may not tolerate SAMe. Rarely nausea or diarrhea becomes severe enough to require discontinuation. SAMe can occasionally cause headaches and exacerbate anxiety, panic, or agitation, as can any activating antidepressant. A diagnosis of bipolar disorder is a strong contraindication to the use of SAMe; antidepressants, including SAMe, can trigger hypomania or mania. In rare cases, patients with preexisting palpitations have complained of increased irregular heartbeats with SAMe.
The quality of the SAMe and the tablet are critical. Low-quality SAMe oxidizes and loses potency over time. Inexpensive and widely sold low-grade SAMe lacks efficacy and may cause adverse effects. Physicians can guide patients to purchase only the best quality SAMe enteric-coated tablets in individual foil blister packs. Best quality formulas and brands are those that have been shown to be effective in clinical trials. In addition, the clinical observations of expert CAIM practitioners are invaluable.
Conclusion
Health care systems are buckling under the strain of the COVID-19 pandemic. Each surge in hospitalizations further exhausts the health care workforce and leaves in its wake millions who are emotionally shaken and traumatized. In addition to conventional therapies, CAIM treatments can help meet the urgent global needs for mental health care.
Physicians can easily gain competence integrating into patient care three CAIM treatments with solid evidence of safety and efficacy—Voluntarily Regulated Breathing Practices, R. rosea, and SAMe—thereby supporting the well-being and stress resilience of their patients, their colleagues, and themselves. ■
References
-
Bottiglieri T. Folate, vitamin B12, and S-adenosylmethionine. Psychiatr Clin N Amer. 2013; 36: 1–14.
-
Bottiglieri T, Gerbarg PL, Brown RP S-Adenosylmethionine, Adometionine, SAMe. In Gerbarg PL, Muskin PR, Brown RP (eds.) Complementary and Integrative Treatments in Psychiatric Practice. Washington D.C., American Psychiatric Association Publishing; 2017: 41-52.
-
Brown RP, Gerbarg PL. The Healing Power of the Breath: Simple Techniques to Reduce Stress and Anxiety, Enhance Concentration, and Balance Your Emotions. Boston, MA: Shambhala Publications; 2012a.
-
Brown RP, Gerbarg PL. Breathing Techniques in Psychiatric Treatment. In Gerbarg PL, Muskin PR, Brown RP (eds.) Complementary and Integrative Treatments in Psychiatric Practice. Washington D.C., American Psychiatric Association Publishing; 2017: 241-250.
-
Brown RP, Gerbarg PL, Muskin PR. How to Use Herbs, Nutrients, and Yoga in Mental Health Care. New York, NY: WW Norton; 2009.
-
Deligiannidis KM, Freeman MP. Complementary and Alternative Medicine for the Treatment of Depressive Disorders in Women. Psychiatr Clin N Amer. 2010; 33: 441-463.
-
Gao L, Wu C, Liao Y, Wang J. Antidepressants effects of Rhodiola capsule combined with sertraline for major depressive disorder: A randomized double-blind placebo-controlled clinical trial. J Affective Dis. 2020; 265:99-103.
-
Gerbarg PL, Brown RP. Mind-Body Interventions to reduce Coronavirus Pandemic Stress and Support Long-Term Recovery. J Acupuncture and Traditional Medicine. 2021, 4(1):1-5.
-
Gerbarg PL, Brown RP. Neurobiology and neurophysiology of breath practices in psychiatric care. Psychiatric Times. 2016a; 33: 22-25.
-
Gerbarg PL, Brown RP. Pause menopause with Rhodiola rosea, a natural selective estrogen receptor modulator. Phytomedicine. 2016b; 23: 763-9.
-
Gerbarg PL, Brown RP, Streeter CC, et. al. Breath Practices for Survivor and Caregiver Stress, Depression, and Post-traumatic Stress Disorder: Connection, Co-regulation, Compassion. Integrative and Complementary Medicine OBM. 2019; 4; 1-24.
-
Gerbarg PL, Jacob VE, Stevens L, et al. The Effect of Breathing, Movement, and Meditation on Psychological and Physical Symptoms and Inflammatory Biomarkers in Inflammatory Bowel Disease: A Randomized Controlled Trial. Journal of Inflammatory Bowel Disease. 215; 21: 2886-96.
-
Gerbarg PL, Muskin PR, Brown RP (eds). Complementary and Integrative Treatments in Psychiatric Practice. Washington D.C., American Psychiatric Association Publishing; 2017.
-
Herrero JL, Khuvis S, Yeagle E, et al. Breathing above the brain stem: volitional control and attentional modulation in humans. J Neurophysiol. 2017; 119: 145–159.
-
Mischoulon D, Price LH, Carpenter LL, et al. Reply to Failed studies should not be used to malign good treatments. J Clin Psychiatry. 2014; 75: 11.
-
Nyer M, Gerbarg PL, Liveri MM, et al. A randomized controlled dosing study of Iyengar yoga and coherent breathing for the treatment of major depressive disorder: Impact on suicidal ideation and safety findings. Complement Ther Med. 2018; 37: 136-142.
-
Porges SW. The polyvagal theory: New insights into adaptive reactions of the autonomic nervous system. Cleveland Clin J Medicine. 2009; 76 Suppl 2, S86-90.
-
Porges SW, Carter SC. Polyvagal theory and the social engagement system. In Gerbarg PL, Muskin PR, Brown RP (eds.) Complementary and Integrative Treatments in Psychiatric Practice. Washington, DC.: American Psychiatric Association Publishing; 2017, 221–240.
-
Sharma A, Gerbarg PL, Bottiglieri T, et al. S-Adenosylmethionine (SAMe) for Neuropsychiatric Disorders: A Clinician-Oriented Review of Research. J Clin Psychiatry. 2017; 78: e656-e667.
-
Streeter CC, Gerbarg PL, Brown RP, et. al. Thalamic Gamma Aminobutyric Acid Level Changes in Major Depressive Disorder After a 12-Week Iyengar Yoga and Coherent Breathing Intervention. Journal of Alternative and Complementary Medicine. 2020; 26: 190-197.
Supplemental References
-
Amirkhan JH. A Brief Stress Diagnostic Tool: The Short Stess Overload Scale. Assessment. 2018; 25: 1001-1013.
-
Beauchaine TP. Future Directions in Emotion Dysregulation and Youth Psychopathology. J Clin Child Adolesc Psychol. 2015; 44: 875-96.
-
Bottiglieri T, Godfrey P, Flynn T, et al. Cerebrospinal fluid S-adenosylmethionine in depression and dementia: effects of treatment with parenteral and oral S-adenosylmethionine. Journal of Neurology, Neurosurgery and Psychiatry. 1990; 53: 1096–1098.
-
Brown RP, Gerbarg PL. The Rhodiola Revolution. Rodale Press, New York. 2004.
-
Brown RP & Gerbarg PL. Non-Drug Treatments for ADHD: New Options for Kids, Adults, and Clinicians. New York, NY: WW Norton. 2012b.
-
Brown RP, Gerbarg PL & Ramazanov Z. A phythomedical review of Rhodiola rosea. Herbalgram. 2002; 56: 40–62.
-
Carrieri PB, Indaco A, Gentile S, et al. S-adenosylmethionine treatment of depression in patients with parkinson’s disease: A double-blind, crossover study versus placebo. Curr Ther Res. 1990; 48: 154-160.
-
Cerutti R, Sichel MP, Perin M, et. al. Psychological distress during the puerperium: a novel therapeutic approach using S-adenosylmethionine. Curr Ther Res. 1993; 53: 701-716.
-
Darbinyan V, Aslanyan G, Amroyan E, et al. Clinical trial of Rhodiola rosea L. extract SHR-5 in the treatment of mild to moderate depression. Nord J Psychiatry. 2007; 61: 343-8.
-
Edwards D, Heufelder A, Zimmerman A. Therapeutic effects and safety of Rhodiola rosea extract WS® 1375 in subjects with lifestress symptoms–Results of an open-label study. Phytotherapy Research. 2012; 26: 1220–1225.
-
Gerbarg PL, Brown RP. Non-Western Interventions for Stress Reduction and Resilience. British Journal of Psychiatric Advances. In press.
-
Gerbarg PL, Brown RP, Mansur S, et al. Survivors of Mass Disasters: Breath-Based Mind-Body Interventions and Global Platforms. In Okpaku, S (ed.) Innovations in Global Mental Health. Switzerland AG: Springer Nature. In press.
-
Gerbarg PL, Brown RP, Muskin PR. Complementary and Integrative Medicine, DSM-5, and clinical decision making. In Gerbarg PL, Muskin PR, Brown RP (eds.) Complementary and Integrative Treatments in Psychiatric Practice. Washington D.C.: American Psychiatric Association Publishing. 2017: 9-20.
-
Gerbarg PL, Wallace G & Brown RP. Mass disasters and mind-body solutions: evidence and field insights. International Journal of Yoga Therapy. 2011; 21: 97–107.
-
Hardy ML, Coulter ID, Favreau JT, et al. S-adenosyl-L-methionine for treatment of depression, osteoarthritis, and liver disease. Evidence Reports/Technology Assessments, No. 64. Rockville (MD): Agency for Healthcare Research and Quality (US); Report No.: 02-E034ISBN-10: 1-58763-122-9. 2002.
-
Hsu S-M, Tseng C-H, Hsieh C-H, et al. Slow-paced inspiration regularizes alpha phase dynamics in the human brain. J Neurophysiol. 2020; 123: 289-99.
-
Hung SK, Perry R, Ernst E. The effectiveness and efficacy of Rhodiola rosea L.: a systematic review of randomized clinical trials. Phytomedicine. 2011; 15;18: 235-44.
-
Koop T, Dienel A, Heldmann M, Münte TF. Effects of a Rhodiola rosea extract on mental resource allocation and attention: An event-related potential dual task study. Phytotherapy Research. 2020; 34: 3287-3297.
-
Kurkin VA, Zapesochnaya GG. Chemical composition and pharmacological characteristics of Rhodiola rosea (review). Journal of Medicinal Plants. Russian Academy of Science, Moscow. 1985; 1231-445.
-
Lekomtseva Y, Zhukova I, Wacker A. Rhodiola rosea in subjects with prolonged fatigue syndromes: results of an open-label clinical trial. Complement Med Res. 2017; 4:46–52.
-
Li Y, Pham V, Bui M, Song L, Wu, C, Walia A, Uchio E, et al. (2017) Rhodiola rosea L.: an herb with anti-stress, anti-aging, and immunostimulating properties for cancer chemoprevention. Curr Pharmacol Rep. 3(6):384-395.
-
Mischoulon D (2009) Update and critique of natural remedies as antidepressant treatments. Obstet Gynecol Clin North Am. 2009; 36: 789-807.
-
Mischoulon D, Alpert JE, Arning E, et al. Bioavailability of S-adenosyl methionine and impact on response in a randomized, double-blind, placebo-controlled trial in major depressive disorder. J Clin Psychiatry. 2012; 73: 843-8.
-
Muskin PR, Gerbarg PL, Brown RP. Along roads less traveled: Complementary, alternative, and integrative treatments. Psychiatric Clinics of North America. 2013; 36: xiii-xv.
-
Panossian A & Amsterdam D. Adaptogens in psychiatric practice. In Gerbarg PL, Muskin PR, Brown RP (eds.) Complementary and Integrative Treatments in Psychiatric Practice. Washington D.C.: American Psychiatric Association Publishing. 2017; 113-134.
-
Panossian A, Wikman G, et al. Rosenroot (Rhodiola rosea): traditional use, chemical composition, pharmacology and clinical efficacy. Phytomedicine. 2010; 17: 481–493.
-
Porges SW. Social engagement and attachment. A Phylogenetic Perspective. Ann NY Acad Sci. 2003: 1008: 31-47.
-
Sanders M, Gerbarg PL. In an insecure world breath-based practices promote attachment, emotional development, and resilience. Presentation at the American Psychiatric Association Annual Meeting Online, May 2021.
-
Saratikov AS, Krasnov EA. Chapter VII: Adaptogenic properties of Rhodiola rosea. In: Saratikov AS, Krasnov (eds). Rhodiola rosea is a valuable medicinal plant (Golden Root). Tomsk, Russia: Tomsk State University Press. 1987: 194-215.
-
Zhou F, Gao B, Wang X, Li J. Meta-analysis of ursodeoxycholic acid and Sadenosylmethionine for improving the outcomes of intrahepatic cholestasis of pregnancy. [Article in Chinese]. [abstract only]. Zhonghua Gan Zang Bing Za Zhi. 2014; 22: 299-304.
Author Disclosures
Patricia L. Gerbarg, M.D., and Richard P. Brown, M.D., receive no financial remuneration from any companies selling or marketing the supplements described in this article. They receive some payments for teaching Breath-Body-Mind practices and for training other teachers. They are members of the Board of the Breath-Body-Mind Foundation, a 501(c)3 not-for-profit. Philip R. Muskin, M.D., M.A., has no activities or interests relevant to the content of the article.
link